Shortened Benznidazole Regimens to improve the efficiency of the treatment for Chagas disease
A study conducted by DNDi researchers will test how to maintain positive clinical results while making the process easier for patients to tolerate and complete
10/10/2025
Chagas disease, or American trypanosomiasis, is a Neglected Tropical Disease caused by the Trypanosoma cruzi. This protozoan parasite can affect patients through several routes, mainly vector-borne transmission through the bite of a triatomine insect, and vertical (congenital) or mother-to-child transmission during pregnancy. Other routes include ingestion of contaminated food and drinks and, less frequently, transfusions from affected donors.
Currently, there are two drugs indicated for the treatment of Chagas disease, Nifurtimox and Benznidazole, which are especially effective when supplied early. However, these drugs can carry adverse effects that could compromise adherence in affected individuals.
NuestroBen study will test reducing the standard eight weeks treatment with Benznidazole to a regimen of two or four weeks, through a randomized clinical trial with 540 participants in centers from Argentina and Bolivia. Recruitment will select T. cruzi-positive individuals aged 18–60 years who have not previously been treated with any of both the drugs. DNDi researchers will evaluate the sustained elimination of parasitemia (blood parasite indicator) from the end of treatment to a maximum of 12 months of follow-up. They will also evaluate whether these shortened benznidazole regimens improve drug tolerability and adherence compared to standard treatment, while maintaining its efficacy (75-80%) in participants with indeterminate Chagas disease or mild cardiac involvement.
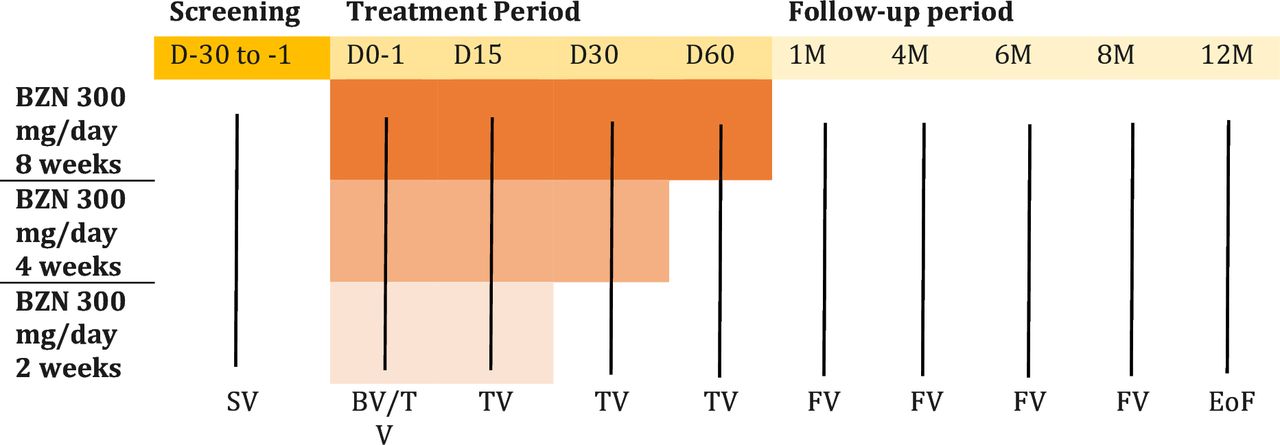
This study was approved in May 2025 by the Universidad Autónoma Juan Misael Saracho (UAJMS) and is funded by the Drugs for Neglected Diseases Initiative (DNDi), ELEA-Phoenix S.A., and the Mundo Sano Foundation.
Read the full article on BMJ Journals
SHARE
Other news